Apple Watch Atrial Fibrillation History Feature Now Available in Mainland China
Apple has officially launched its Atrial Fibrillation (AFib) History feature for the Apple Watch in mainland China, a significant development for cardiovascular health monitoring in one of the world’s most populous countries. This rollout, confirmed on Tuesday, March 17, 2026, by Apple’s China Newsroom, brings a sophisticated health tracking capability to a new, vast user base. The feature, first introduced in the United States in 2022 with watchOS 9, allows individuals diagnosed with atrial fibrillation to gain a more detailed understanding of their condition by estimating the frequency of irregular heart rhythms.
Understanding Atrial Fibrillation History
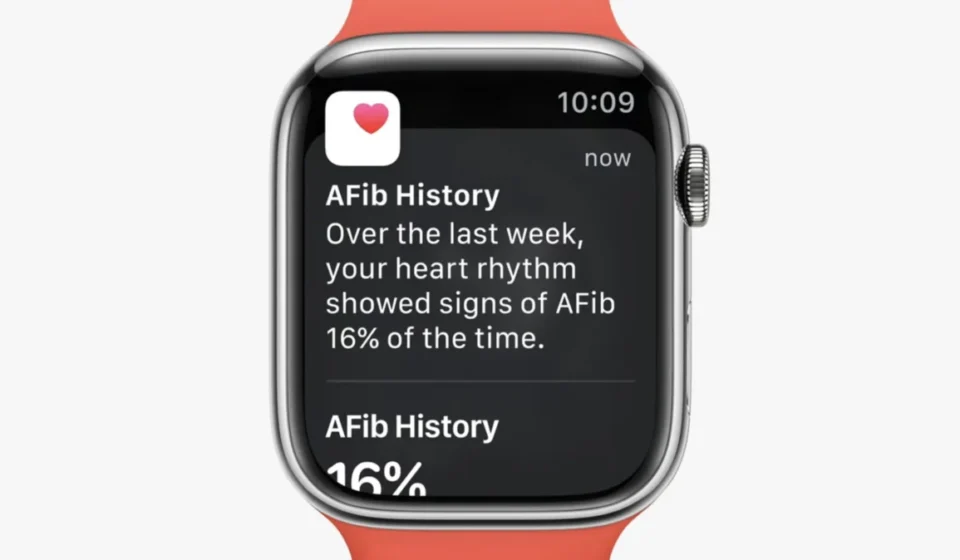
The AFib History feature operates by leveraging the Apple Watch’s photoplethysmography (PPG) sensor, which monitors pulse rate. Through advanced algorithms, the device analyzes this data to identify patterns consistent with atrial fibrillation. The core output of the feature is a retrospective "AFib burden" estimate. This metric quantifies the percentage of time the user’s heart rhythm has been identified as AFib during periods when the Apple Watch was worn. Apple specifies that this feature is intended for use by individuals aged 22 and older who have received a formal diagnosis of atrial fibrillation from a qualified physician. It is crucial to note that the feature is designed as a supportive tool for managing a diagnosed condition, not as a primary diagnostic instrument for those without a prior medical assessment.
A Phased Global Expansion and Regulatory Hurdles
The introduction of AFib History in mainland China follows a strategic global expansion of the Apple Watch’s advanced health features. Since its initial release in the U.S. in 2022, Apple has progressively made this and other cardiovascular health functionalities available in numerous regions worldwide. Features such as the Electrocardiogram (ECG) app and Irregular Heart Rhythm Notifications have become staples for Apple Watch users in over 150 countries.
However, the timeline for the availability of these medical-grade features can vary significantly by region due to diverse regulatory landscapes. In China, health-related technologies, particularly those classified as medical or quasi-medical functions like ECG and AFib tracking, require stringent approval from the National Medical Products Administration (NMPA). The NMPA’s rigorous evaluation process is designed to ensure the safety, efficacy, and reliability of medical devices and software. The delay in launching AFib History in mainland China is directly attributable to navigating these complex regulatory requirements. Apple has historically maintained a comprehensive list of Apple Watch feature availability on its official website, which is regularly updated to reflect regional rollouts and approvals.
Background: The Evolution of Cardiovascular Health on Apple Watch
The journey towards advanced cardiovascular monitoring on the Apple Watch began years before the AFib History feature. Apple’s commitment to integrating health and wellness into its ecosystem has been a cornerstone of its product strategy.

- Early Innovations: The Apple Watch Series 1, released in 2015, laid the groundwork for health tracking with features like activity monitoring and heart rate sensing.
- Introducing ECG: A significant leap occurred with the Apple Watch Series 4 in 2018, which introduced the ECG app, enabling users to take an on-demand electrocardiogram. This feature received FDA clearance in the United States, marking a pivotal moment for consumer wearables in the medical field.
- Irregular Rhythm Notifications: Following the ECG app, Apple introduced Irregular Heart Rhythm Notifications, which could periodically check a user’s heart rhythm in the background and notify them if it suggested signs of AFib. This proactive monitoring capability aimed to alert users to potential issues even when they weren’t actively seeking a diagnosis.
- AFib History: The AFib History feature, introduced in watchOS 9 in 2022, represented a further refinement, providing users with a more quantifiable understanding of their AFib patterns over time. This added layer of data empowers both users and their healthcare providers to make more informed decisions regarding treatment and lifestyle adjustments.
The introduction of AFib History in China signifies Apple’s ongoing efforts to democratize access to advanced health monitoring tools, adapting its technology to meet the specific needs and regulatory frameworks of diverse global markets.
Data and Potential Impact in China
The introduction of the AFib History feature holds considerable potential for China, a nation grappling with a significant burden of cardiovascular diseases. According to the World Health Organization (WHO), cardiovascular diseases are the leading cause of death globally and in China. Atrial fibrillation, in particular, is a major risk factor for stroke, heart failure, and other serious cardiovascular complications.
- Prevalence of AFib: While precise, up-to-date figures for AFib prevalence specifically in mainland China can be challenging to pinpoint, global estimates suggest a significant portion of the adult population is affected. Factors such as an aging population, rising rates of hypertension, diabetes, and obesity contribute to an increasing prevalence of AFib.
- Empowering Patients: By providing individuals with data on their AFib burden, the feature can foster greater engagement in their own health management. This increased awareness can motivate users to adhere to prescribed treatments, adopt healthier lifestyles, and communicate more effectively with their doctors.
- Early Detection and Intervention: For individuals at risk, or those with a known diagnosis, the Apple Watch can serve as a valuable tool for monitoring. Early detection of increased AFib burden could prompt timely medical intervention, potentially reducing the incidence of stroke and other AFib-related complications.
- Contribution to Public Health Data: As more users adopt the feature, the anonymized and aggregated data collected could potentially contribute to a broader understanding of AFib patterns within the Chinese population, aiding in public health research and policy development.
Analysis of the Regulatory Approval Process
The NMPA’s role in approving health-related features for consumer devices underscores the evolving landscape of digital health. The rigorous nature of these approvals reflects a commitment to patient safety and the integrity of medical data.
- Validation and Verification: The NMPA likely conducted thorough reviews of the Apple Watch’s PPG sensor accuracy, the algorithms used for AFib detection, and the overall reliability of the AFib History feature. This validation process ensures that the data presented to users is scientifically sound.
- Data Privacy and Security: As with any health technology, data privacy and security are paramount. Apple’s compliance with China’s stringent data protection regulations would have been a critical component of the approval process.
- Market Entry Strategy: For global technology companies, understanding and navigating the regulatory pathways of major markets like China is crucial for successful product launches. Apple’s consistent approach to securing these approvals demonstrates its long-term commitment to these regions.
Broader Implications and Future Outlook
The availability of AFib History in mainland China is more than just a feature update; it represents a significant step in the integration of consumer technology with personal healthcare.
- Digital Health Ecosystem: This launch further solidifies the role of wearables as integral components of the burgeoning digital health ecosystem. As these devices become more sophisticated and gain regulatory approval, they are poised to play an increasingly vital role in preventative care and chronic disease management.
- Apple’s Health Ambitions: For Apple, this expansion aligns with its broader vision of empowering individuals to take proactive control of their health. The company has consistently invested in research and development to enhance the health and wellness capabilities of the Apple Watch, positioning it as a leading device in the health tech space.
- Competitive Landscape: The move is also likely to influence the competitive landscape of wearable technology and digital health solutions in China. Other technology companies and health service providers may accelerate their own efforts to introduce similar medically validated features.
- Future Feature Rollouts: It is reasonable to anticipate that other advanced health features from Apple, such as enhanced sleep tracking, blood oxygen monitoring, and potentially future innovations in areas like non-invasive glucose monitoring (if developed and approved), will follow a similar path to market in China, subject to regulatory review.
The launch of AFib History in mainland China signifies a harmonious convergence of technological innovation, regulatory diligence, and a growing global demand for accessible health monitoring tools. This development is poised to benefit millions of individuals, offering them a more informed and proactive approach to managing their cardiovascular well-being.







